Er lumen positive charged8/27/2023 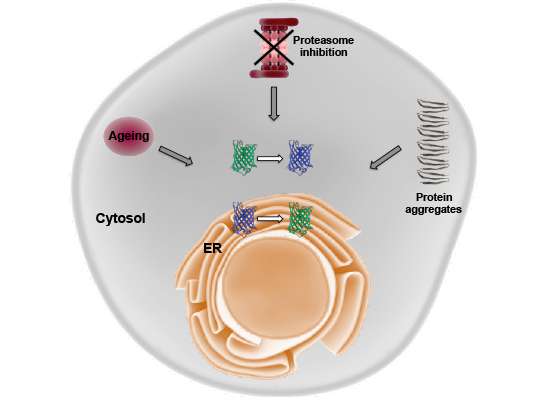
In some cases, the overall conformation of a transmembrane (TM) region rather than a sequence feature is recognized ( Gardner and Hampton, 1999 Sato et al., 2009). For ERAD-M substrates, much remains unknown about the structural features that target proteins for degradation. In mammals, an even more complex proteostasis network including several additional ERAD E3 ligases and auxiliary factors selectively target ER proteins into several parallel degradation routes ( Christianson and Ye, 2014 Mehrtash and Hochstrasser, 2018). At the inner nuclear membrane, a sub-area of the ER, the Asi complex targets mislocalized membrane proteins for proteasomal degradation ( Foresti et al., 2014 Khmelinskii et al., 2014). In yeast, three main E3 ubiquitin ligases target proteins from the ER for proteasomal degradation: Hrd1 is known to recognize ERAD-L and most ERAD-M substrates, whereas ERAD-C substrates are handled by Doa10 ( Carvalho et al., 2006 Vashist and Ng, 2004). Depending on the location of the lesion or the signal for regulated degradation (degron) in the protein, ERAD substrates are classified into three main categories: ERAD-L (lumen), ERAD-M (membrane) and ERAD-C (cytosol). However, the major clearance pathway for misfolded proteins relies on the ER-associated degradation (ERAD) pathway that targets proteins for degradation by the cytoplasmic proteasome ( Christianson and Ye, 2014 Mehrtash and Hochstrasser, 2018 Ruggiano et al., 2014).ĮRAD is commonly defined by its role in eliminating aberrant proteins that could cause harm to the cell. The latter mainly targets terminally misfolded proteins and large substrates whose degradation occurs via selected autophagy or direct ER-to-lysosome trafficking ( Fregno and Molinari, 2019). Here, post-translational control of protein abundance is mediated either by proteasomal degradation or the lysosomal system. The endoplasmic reticulum (ER) is the major site for protein synthesis, maturation and quality control for all proteins within the secretory pathway ( Ellgaard et al., 2016). When not corrected by the protein degradation machinery, imbalance of protein stoichiometry generates a major fitness defect by leading to toxic protein species and protein aggregation ( Brennan et al., 2019). Moreover, a dynamic protein homeostasis (proteostasis) network ensures that damaged cellular proteins are efficiently removed and that protein complexes assemble in the correct stoichiometry ( Juszkiewicz and Hegde, 2018). The later in the biosynthesis pathway regulation takes place, the faster it allows the cell to respond to current demands. This occurs both at the transcriptional and translational level by affecting synthesis and degradation rates of both mRNA and proteins to tightly control the total amount of any protein ( Vogel and Marcotte, 2012). In order to grow, divide and survive under changing conditions, cells need to finely tune the amount of each of their proteins by regulating synthesis, modification and degradation. Control of the cellular proteome is an essential process in every cell.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
